 |
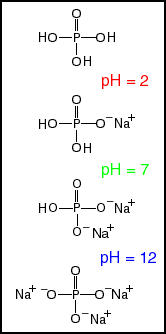
Four forms of phosphate Phosphate exists as a mixture of four species, with relative amounts dependent on the pH of the aqueous environment. At the lowest pH, (most acidic), the predominant form is phosphoric acid. This acid is moderately strong, stronger than acetic or citric acid (the acids in vinegar and lemons), but much weaker than sulfuric acid. Phosphoric acid gives a characteristic taste to soft drinks, e.g. Coke. At pH 2, there is an equal amount of phosphoric acid and ionized phosphate. In the Figure we have sodium as the counter ion. At pH 7, the approximate pH in living cells, there is an equal amount of mono- and di- sodium phosphate. Phosphate buffers containing these two phosphate salts (or equivalents) are frequently used in biochemical and physiological studies. Tri-sodium phosphate (TSP), is quite basic, and is the major species above pH 12. TSP is used as a cleaning agent, because the high pH converts water insoluble fats and oils into water soluble soaps. |
||||||
|
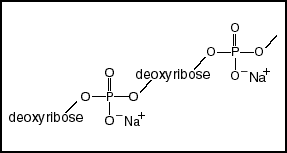
The phosphodiester backbone Acids react with hydroxyl groups to form esters. In DNA each phosphate has formed an ester with a hydroxyol group on two sugars. Each sugar, a deoxyribose, is also linked to two phosphates. This chain, alternating phosphates and sugars, is called the phosphodiester backbone of DNA. While in this diagram the backbone looks symmetrical, when we look at the structure of the deoxyribose, we will see that it has a polarity. |
|||||||
 |
|||||||
| The single remaining charged oxygen on the phosphates makes DNA an acid (the A in DNA). These negative charges would strongly repel each other and destabilize the DNA molecule, except for the electrostatic screening by positive counter ions, mostly sodium, potassium, and magnesium inside the cell. | |||||||